Converts clinical practice into analysable data through disease registries, protocol documentation, case reports, case series, clinical audits, publications and long-term outcome monitoring.
Call : +91 485 225 3007
email : support@sreedhareeyam.com
Experts chart roadmap for integrated diabetic retinopathy management at 'Sunetra 2026'
Converts clinical practice into analysable data through disease registries, protocol documentation, case reports, case series, clinical audits, publications and long-term outcome monitoring.
Supports objective evaluation with refraction, tonometry, slit-lamp examination, retinoscopy, lensometry, visual field analysis, OCT, B-scan and fundus photography.
Assists systemic assessment, safety monitoring and research involving diabetes, hypertension, inflammation, autoimmune-associated ocular disease and metabolic disorders.
Strengthens medicine standardisation through pharmacognostic analysis, physicochemical testing, phytochemical screening, HPLC, HPTLC, heavy metal analysis, microbiology and stability studies.
Connects research with outpatient care, inpatient care, Ayurvedic procedures, pharmacy, diet support, nursing care, follow-up systems and specialist expertise.
Uses electronic records, ophthalmic imaging, laboratory reports, video consultation support, patient portals and digital follow-up to reduce data loss and improve continuity.
Extends screening, treatment and documentation to underserved patients through the service principle of Vision for All.

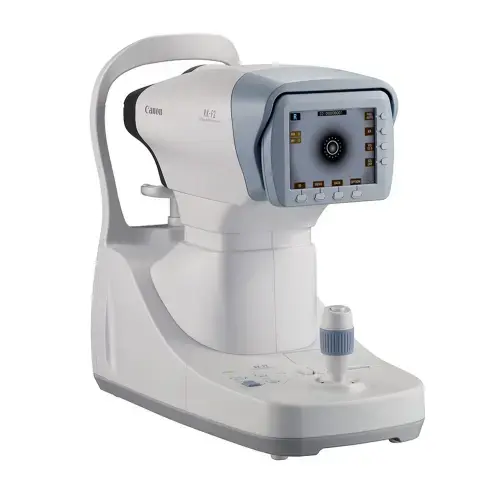
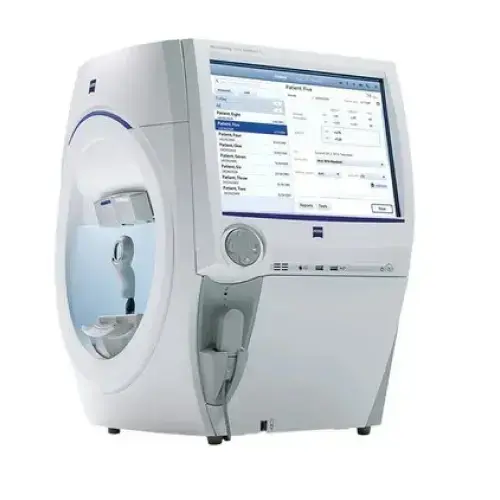
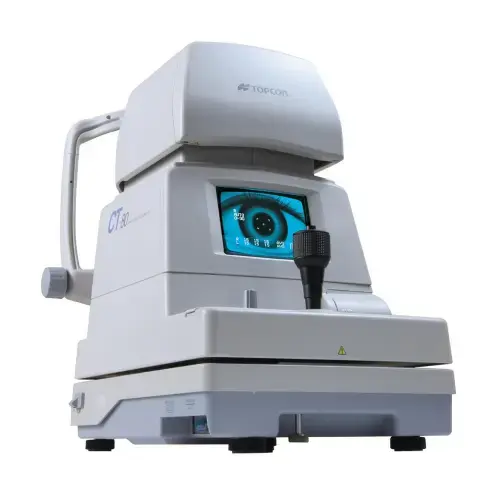
Modern diagnostic tools support clinical evaluation and research documentation before, during and after treatment.
We use cookies that are necessary for the smooth operation of the website, to improve our website and to display advertising relevant to you on social media platforms and partner websites. By clicking "Accept all", you agree to the use of cookies for convenience features and statistics and tracking. You can change these settings again at any time. If you do not agree, we will limit ourselves to technically necessary cookies. For more information, please see our privacy policy .